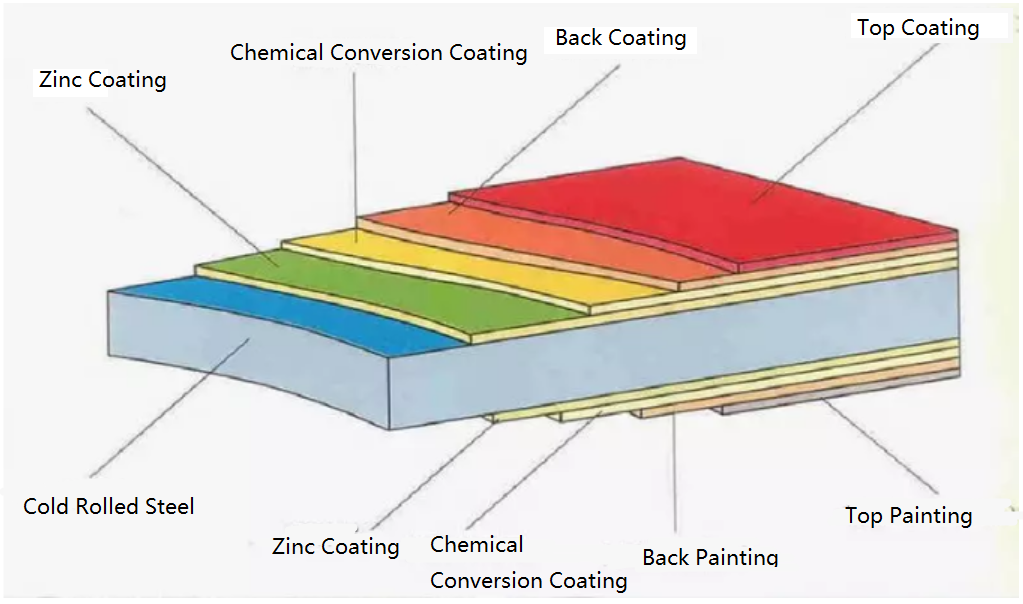
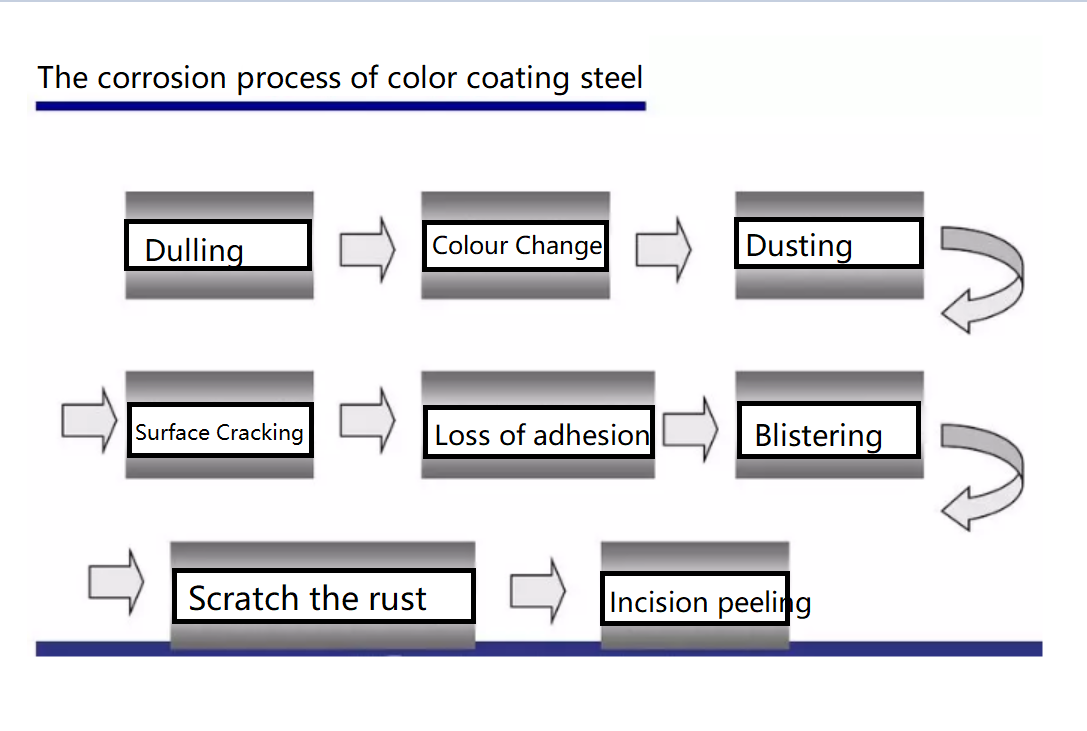
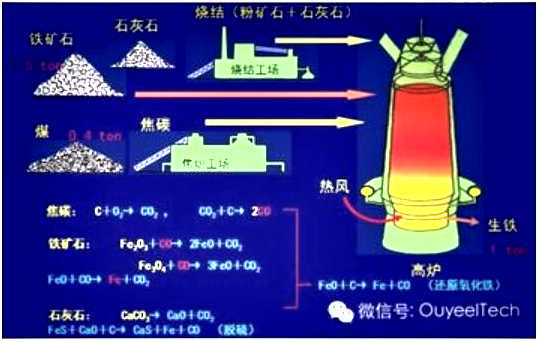
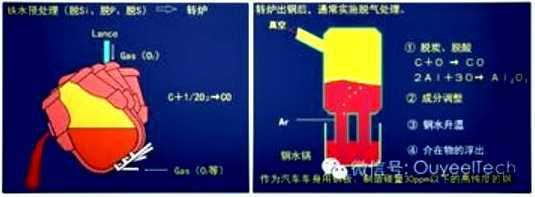
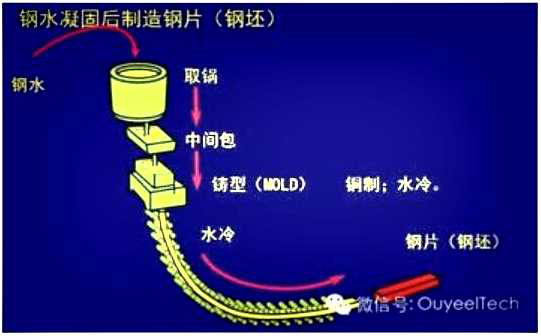
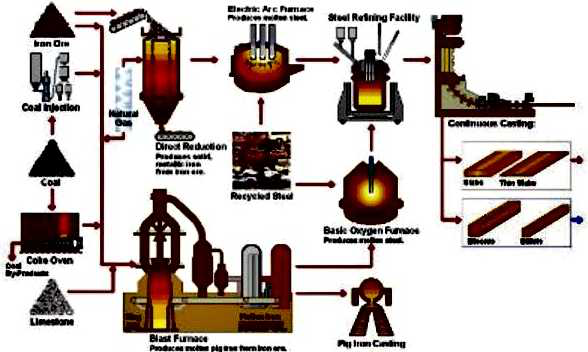
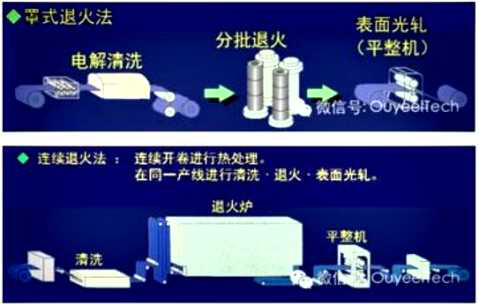
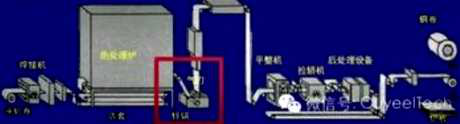
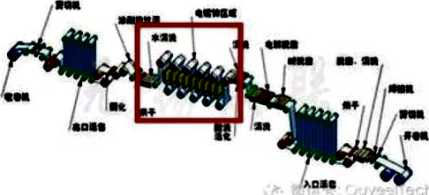
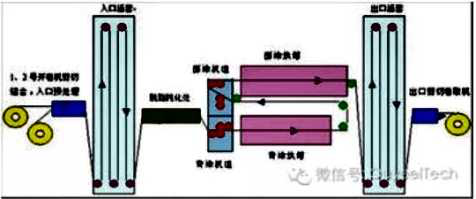
Hot dip galvanizing is one of the effective methods to improve the corrosion resistance of steel because of its excellent coating performance and long service life.
The surface of hot dip galvanizing layer has many common quality defects such as leakage plating, stripe feather white pattern, coating super thick, rough, gray, tear streaks, turtle crack, crystal raised grain, “white rust”, etc., the influencing factors are many, and the control is relatively difficult.
In order to improve the surface quality of hot dip galvanizing, the process control should be strengthened and necessary measures should be taken.
Surface pretreatment
Poor surface pretreatment is the main cause of plating leakage.
Steel in the factory, storage, transportation, processing inevitably stained with paint or mineral fat, sometimes welding parts need to daub difficult to clean special grease, most of the enterprise and no degreasing process, only by pickling to remove surface impurities, which is easy to cause leakage plating (iron).
The other case is due to the pickling process pickling parts more concentrated, piled up dense, heavy and other factors, resulting in underpickling and line clamping, or because the pickling concentration is too high so that the acid salt precipitation on the surface, groove, not washing or washing is not complete, it is also easy to form leakage plating, empty plating phenomenon.
In addition, due to solvent aging, failure, in addition to the drying process did not put the workpiece in time, or drying time is longer so that the steel surface appears secondary micro-oxidation, the formation of acid iron salt cover on the surface, also prone to plating leakage, especially in the relative humidity of the climate and fog weather is more common.This situation is often overlooked.
In addition, when using solvent hot dip galvanizing, the most commonly used ZnCI2 and NH4CI mixed aqueous solution is not the right ratio, no eutectic point is formed, especially in the case of too high concentration, in the support plate welding Angle seam circular hole is prone to occurrence of ash trap leakage plating.
The zinc coating is not thick enough
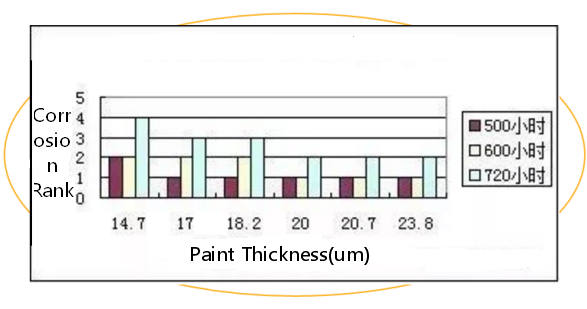
The thickness of galvanized layer is not up to the standard requirements will directly affect the service life of the product.The reasons for the insufficient thickness of the galvanized layer are as follows.
(1)Effect of zinc liquor temperature.When the temperature of zinc solution is lower than 430°C, the diffusion rate of zinc and iron is low, it is not easy to generate enough iron and zinc alloy layer, the whole coating is thin;When the temperature is 460°C, the zinc layer thickens; when the temperature continues to rise, the zinc solution becomes thinner and the zinc layer becomes thinner.Therefore, the temperature of zinc liquid must be strictly controlled to ensure the stability of quality.
(2) influence of dwell time of strip steel in zinc solution.The dwell time of strip steel in zinc solution has a certain time range.According to the thickness of strip steel, with the extension of galvanized time, the coating thickness will increase, but excessively prolonged galvanized time will make the zinc layer brittle, but affect the quality.
(3) influence of zinc extraction method from zinc solution.The zinc coating should be thin when the strip is slanted out.Therefore, when the strip leads out the zinc pot, it should be vertical.
Other components in zinc solution
The zinc solution contains many other metal components or harmful elements, which may cause defects such as zinc slag particles adhering to the surface of the coating and some abnormal patterns, cracks and other defects.
(1) iron
Hot dip galvanized for a period of time, the steel surface will appear zinc accumulation and small zinc slag particles, resulting in the coating surface rough, smooth also declined.Such fine particles are generally Fe – Zn alloy particles.
At 450 ° C galvanized, zinc iron in the saturated solubility of 0.02%, iron content in liquid zinc exceeds this value, can form zinc slag, due to the difference of zinc slag particles and density is not large, the gathered sinking slowly, suspended in the liquid zinc alloy slag will clip in zinc liquid out of the parts, inlaid in the coating, which affect the quality of the galvanized layer surface.The sources of iron in zinc solution are generally the corrosion of zinc pot, the dissolution of the parts, the iron ions in the plating agent and the iron salts on the parts.
Therefore, in order to obtain a flat and smooth coating, it is necessary to strictly control the iron content in the zinc solution, reduce the iron ion, control the temperature of the zinc solution, to avoid the zinc solution temperature of the rise and fall, reduce the corrosion rate of the zinc pot.Generally, when the content of iron in zinc solution should be 0.20%, it is necessary to cool down and stand still and salvage zinc slag.
(2) aluminum
Aluminum is the most commonly used additive element in hot dip galvanizing.Different properties of zinc coating can be obtained by adding different concentrations of aluminum to zinc solution.
It is generally believed that hot galvanizing, adding < 1% aluminum in the zinc solution can play the following role: improve the coating brightness;Reduce the oxidation of zinc liquid surface;The coating with good adhesion was obtained by inhibiting the formation of brittle Fe – Zn phase.
However, in the actual production, the zinc solution containing 0.005%~0.020%AI can achieve the purpose of bright coating, and can reduce the surface oxidation of zinc solution and the generation of zinc ash.
It must be pointed out that Zn – AI intermediate alloy should be used to add aluminum.Often due to add the quality problems in the middle of the alloy and the method of adding aluminum and zinc alloy inappropriate or too fast, leading to a large number of generated in zinc liquid aluminum, iron compounds, the density of liquid zinc is less than the zinc aluminum iron three yuan “surface slag” or zinc particles suspended in a liquid surface and very thick, very easy to stick on the iron and steel parts, serious damage to the coating quality, only by cooling in dregs is useless at this time.
Once found this situation should immediately stop adding alloy, at the same time to take zinc solution purification measures, aluminum content down, when the situation is not serious can consider the appropriate extension of air cooling time, in order to prevent the appearance of bubbles and wrinkles coating surface sharp water cooling.
(3) the tin and lead
Generally, zinc ingots contain no tin, only trace amounts of lead.In recent years, some suppliers of zinc alloy have added tin and lead to the so-called multi-alloy in order to reduce the temperature of zinc plating and obtain white and bright coatings.
When adding the liquid zinc alloy, zinc liquid level can appear mirror under 430 ° C and can maintain 10 ~ 20 min, but in hot steel plated parts, the coating on the surface of the plume tinea and small white spot, tin, lead alloy added content once reached 0.5%, zinc flower will occur and zinc ash significantly raised, production efficiency is also decreased, caused losses to the enterprise.
Lead and tin, due to their low melting point and high content, rapidly reduce the solidification temperature of the zinc solution. During the cooling process of the zinc layer, it is easy to form coarse crystals and turtle cracks, which affect the surface smoothness and corrosion resistance of the zinc layer.
Intergranular corrosion is the most sensitive to aluminum impurities, and the corrosion rate is accelerated with the increase of lead content in the coating. When the lead content reaches 0.02%, intergranular corrosion will occur. The coating will break along the grain boundary and lose adhesion.Therefore, we must make clear the content of lead, tin and aluminum in the application of zinc-aluminum alloy or multiple alloy, and decide whether to add it in the zinc solution.
Galvanized process
The main problems of the imperfect hot-dip galvanizing process are as follows:
(1) after pickling, no washing or insufficient washing can directly enter the solvent pool, so as to increase the acidity of the solvent and increase the iron ion;Or with hydrochloric acid as a plating agent, so that the strip steel surface covered with a layer of iron salt, into the zinc solution, the formation of melting slag attached to the surface of the workpiece;
(2) solvent aging, more iron ions, more impurities, if not filtered or with the strip into the zinc solution, the viscosity of the zinc solution increased, making the coating thicker and rough, poor adhesion;
The workpiece does not dry, the solvent with water for hot galvanizing will form zinc (0H)2 particles, affecting the product appearance quality;
(4) the solvent is generally weak acid, no heating or low temperature will prolong the drying time, will also corrodes the surface of the workpiece and form adhesion of iron salt, zinc alloy slag formed on the surface of the workpiece, so that the surface of the coating rough
“White rust” is the galvanized layer of the common defects, the main reason is because the zinc plating passivation of the lack, strip with water or do not remove passivation passivation technology is unqualified, strip after water-cooling not completely dry, stored in a damp and electrochemical corrosion in the poorly ventilated environment “white rust” and gray powder corrosion deposits, a direct impact on product and engineering quality, type of this kind of white or grey powder if owner by alkali carbonate zinc compounds, etc
In a word, after hot galvanizing strip steel surface defects are more complex, the causes are more, the solution is a variety of, need to do a lot of hard if meticulous work, so that the hot plating technology to improve one step.